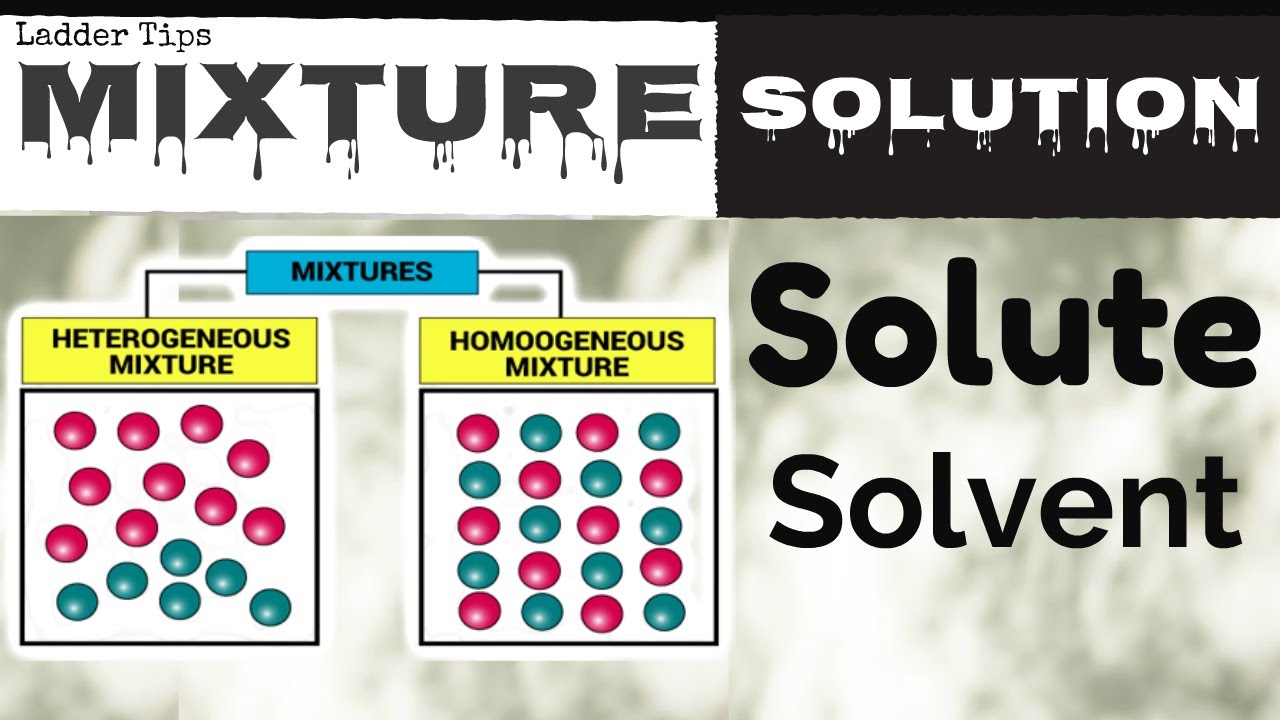
Two or more atoms (same or different) may join together forming molecules (p.12) Atoms may combine with other atoms to form larger particles, called moleculesġ00 different elements = 100 different atoms Atoms of one element are like one another and are different from atoms of all other elementsģ. All matter is made up of tiny particles called atomsĢ. For example, atoms are too small to see so scientists develop models to explain the behavior of matter.ġ. atoms at higher temperatures move faster on average than particles at a lower temperature.Ī scientific model help us visualize process that cannot be seen directly.each pure substance has its own kind of atoms, different from the atoms of other pure substances.all matter is made up of tiny particles called atoms.Mechanical Mixtures - can see different parts of mixtureElements - pure substances that cannot be broken down If a suitable emulsifying agent is added to oil and vinegar salad dressing, the oil droplets cannot clump together to form a separate layer. However, the two liquids quickly separate into layers of oil and vinegar.Įmulsifying agents may keep a liquid dispersed in another liquid. Example, shaking vinegar and oil together to form a salad dressing creates an emulsion. Suspensions can be separated by filtering and light is scattered as it passes through a suspension.Ĭolloids are heterogeneous mixtures composed of fine particles evenly distributed throughout a second substances.Įmulsions such as mayonnaise, milk, and salad dressings are colloids in which liquids are dispersed in another liquid. Example, if you mix flour in water eventually the flour will settle to the bottom of a glass. Suspensions are heterogeneous mixtures made of large particles that are uniformly mixed but will settle if left undisturbed. Ordinary mechanical mixtures, the different particles are big enough to see, and they stay mixed.Įxample - granite is a rock that has different sized particles of quartz, feldspar, and mica. Heterogeneous (mechanical) mixtures are classified according to their particle size. Heterogeneous mixtures are ones that are not uniformly scattered. Light does not scatter when you shine it through a homogenous mixture.Įxamples hydrochloric acid, white vinegar These are usually clear if they are liquids and cannot be filtered to separate particles. Homogeneous mixtures are ones that have their particles uniformly scattered. Mixtures can be classified by their properties. Pure Substance - definite properties always the sameĬompounds - contain 2 or more elements in a fixed proportion (amount) Mixture - contains at least two substances: 3)ĭensity - mass of a specific volume of a substance (example: g/cm3) 
We can classify matter based on different properties - characteristics describe different substances (pg. 
Properties - Those properties we can determine by using our senses an not requiring chemical analysis are called physical properties of matter. Gas - has no definite size, takes shape of container. Liquid - has definite size, takes shape of container 
Matter - Matter is anything that has mass and takes up space. Chemistry is the study of the properties of matter and the changes matter undergoes. dyes and medicines) formed a foundation for the science of chemistry.Ĭhemistry - The science concerned with the properties of matter and changes in matter. In addition, their knowledge of chemicals (i.e. Although they incorporate many superstitious beliefs, their work developed many lab procedures and apparatus we use in chemistry. They also strived to find the Elixir of Life (Fountain of Youth). It was practiced by early Greek and Egyptian metal workers whose goal was to find a magical stone that would turn any ordinary metal into gold. This was the beginning of obtaining knowledge about matter. identify conditions under which properties of material are changed, and critically evaluate if a new substance has been produced identify and apply other methods of classification distinguish between pure substances, solutions, and mechanical mixtures describe and apply different ways of classifying materials based on their composition and properties investigate and describe the properties of materials Investigate materials and describe them in terms of their physical and chemical properties
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
